Bacteria, some of the smallest yet most resilient living organisms on our planet, have evolved a range of strategies to protect themselves from environmental threats and host defenses. This article examines a particularly significant mechanism: the production of capsular polymers. These structures not only serve to shield pathogenic bacteria from desiccation and physical stress, but they also render these microorganisms nearly invisible to the human immune system. Understanding how these capsules form is critical for developing new pharmaceutical interventions.
Capsular polymers are essentially sugar-based shells that encase certain types of bacteria, providing them with crucial survival advantages. These polymers prevent excessive dehydration and shield the bacteria from various physical stresses. However, their most insidious function lies in their ability to camouflage the pathogen from immune responses. By effectively cloaking themselves, these bacteria can persist in hostile environments, such as the human body, for extended periods.
Current research indicates that inhibiting the formation of these capsules could significantly weaken bacterial pathogens, making the enzymes responsible for their production prime targets for drug development. Such advances could pave the way for innovative vaccination strategies and therapeutic agents, potentially altering the landscape of bacterial infection treatment.
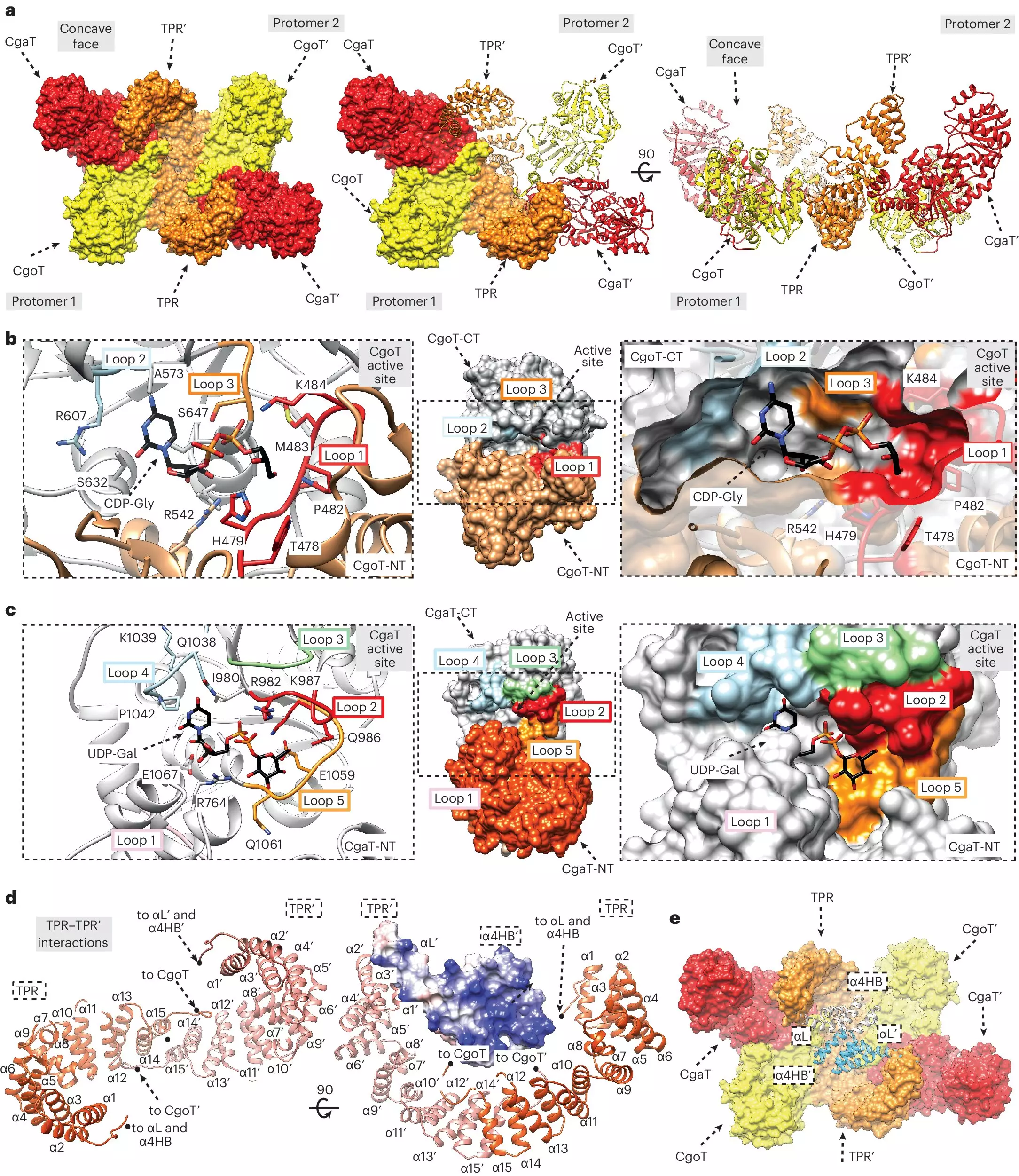
In the ongoing quest to understand bacterial capsule synthesis, a breakthrough has emerged from an international research collaboration led by Dr. Timm Fiebig. This team successfully identified and characterized the enigmatic “linker” that connects the capsular polymers to the bacterial membrane. This discovery is vital, as the linker plays an essential role in the structural integrity of the capsule, serving as an anchor point.
Dr. Fiebig and his team found that the membrane anchor comprises a fatty acid molecule, a crucial component that allows the capsule to adhere to the bacterial cell surface. Prior to this study, the biochemical details surrounding this connection had remained a mystery, limiting our understanding of how capsules are synthesized and, therefore, how they might be targeted.
The study elaborated on the role of transition transferases—enzymes that construct the linker—as major players in the capsular biosynthetic pathway. By isolating and examining these enzymes, the researchers were able to elucidate how they influence the length and structure of sugar chains in the capsule. Notably, a specialized chromatography device helped the researchers reproduce the capsule synthesis in vitro, showcasing how transitional transferases interact with capsular polymerases.
These insights reveal that longer sugar chains, stimulated by transition transferases, contribute to enhanced protective capabilities of the bacterial capsule. This discovery underscores the enzyme’s potential as a target for antibacterial drug design, as inhibiting their activity could compromise the defensive barriers of pathogenic bacteria.
Beyond the immediate implications for antibacterial therapies, this research has significant ramifications for vaccine development. Understanding the genetic makeup and structural nuances of these enzymes can facilitate the design of more effective vaccines. For instance, the findings suggest that transition transferases are conserved across various bacterial genomes, indicating potential common pathways that could be exploited in immunization strategies.
Furthermore, discrepancies between the structural characteristics of the linker and the capsular polymer have prompted researchers to rethink preconceived notions about how these structures develop in bacteria. This fundamental knowledge not only enhances our understanding of microbial biology but also sets the stage for identifying additional enzyme candidates involved in linking the bacterial membrane and capsule.
It is particularly noteworthy that different bacterial species utilize similar methods for synthesizing their capsule-linked structures. This commonality offers a promising avenue for developing broad-spectrum drugs that might operate against multiple bacterial strains. The researchers found parallels among pathogens that cause severe illnesses, such as meningitis and urinary tract infections.
By utilizing enzyme inactivation strategies targeting the linker and capsule formation, we could devise methods to leave bacteria vulnerable to immune response. Ultimately, these findings not only advance our basic understanding of bacterial resilience but could pave the way for innovative therapeutic approaches to combat antibiotic resistance, a pressing issue in contemporary medicine.
Ongoing research into the biochemical pathways of bacterial capsules holds tremendous potential. By elucidating how these protective structures form, scientists are gaining valuable insights that could inform future vaccines and therapeutic strategies against persistent bacterial infections.

Leave a Reply