In the face of climate change and dwindling resources, scientists are turning their attention toward transforming waste molecules into valuable materials. The journey toward achieving meaningful sustainability is long and complex, yet groundbreaking innovations hint at a promising future. Recently, a remarkable international collaboration has emerged, harnessing electricity to refine the process of converting carbon dioxide—traditionally viewed as a harmful greenhouse gas—into methanol, a potent liquid fuel. The implications of this research, published in the prestigious journal Nature Catalysis, could profoundly alter our understanding of waste utilization and renewable energy.
A Glimpse at the Cutting-Edge Research
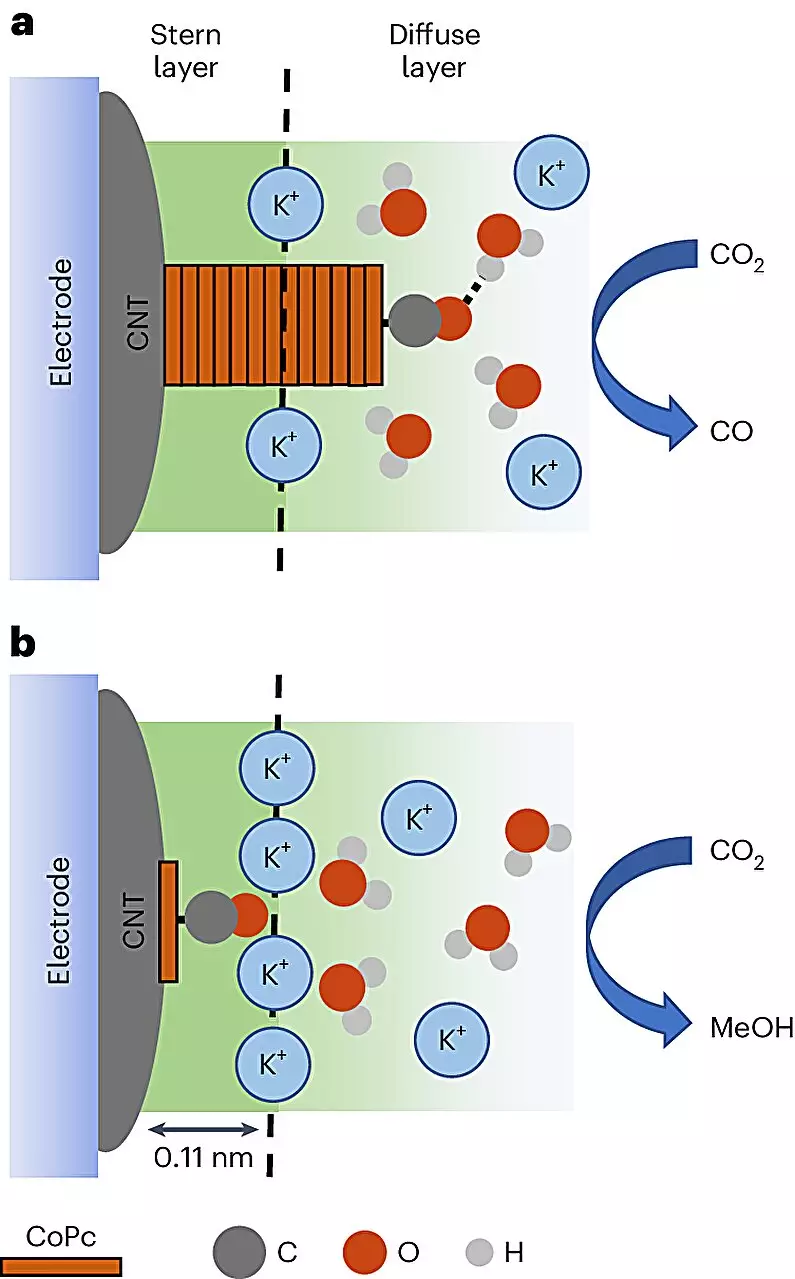
At the heart of this study lies cobalt phthalocyanine (CoPc), a molecule pivotal to achieving a highly efficient conversion of carbon dioxide into methanol. By embedding these molecules into a matrix of carbon nanotubes—remarkable structures known for their exceptional electrical properties—scientists have facilitated a more efficient reaction procedure. The introduction of an electrolyte solution, combined with the application of electrical currents, allows CoPc to absorb electrons from its environment, thereby powering the conversion process. What’s truly fascinating is how this innovative method enables researchers to visualize reactions in real time, revealing critical insights into the behavior of molecules during transformation.
Lead author Quansong Zhu highlights the groundbreaking aspect of vibrational spectroscopy used in this research, a technique that has enabled scientists to observe the distinct changing states of molecules under varying reaction environments. For the first time, they have gathered empirical evidence connecting specific reaction environments to enhanced methanol production. Such advancements provide a much-needed roadmap for future studies aimed at optimizing catalytic reactions while fine-tuning the synthesis of high-value materials.
The Role of Environment in Chemical Reactions
One of the study’s key revelations is the importance of environmental conditions in determining the fate of carbon dioxide during the catalytic process. Within a carefully constructed setting, researchers learned that the same CoPc molecules displayed vastly different outcomes based on their interaction with their immediate environment, specifically showing a pronounced tendency to favor methanol over less desirable by-products like carbon monoxide. The chance for methanol production soared by a staggering factor of eight, showcasing the importance of meticulous control in chemical processes.
This demonstrates a critical realization: to refine catalytic processes, scientists must delve deeper into the nuanced interplay between catalysts and their surroundings. “When we optimize catalysts, we must fully understand the intricacies of their interactions and mechanisms,” argues Robert Baker, co-author of the study and esteemed professor of chemistry at The Ohio State University. Such exploration is essential to unraveling the complexities inherent in catalysis, a field often obscured by monumental layers of empirical findings.
Discovering the Cation Connection
Even more intriguing is the study’s discovery regarding cations—positively charged ions that interact directly with the molecules engaged in the catalytic process. Drawing a parallel with past research, Baker emphasizes the need for further investigations into cation behavior and its role in facilitating faster and more efficient methanol production. The discovery that these cations enhance the conversion processes cultivates excitement for what lies ahead in catalysis and renewable fuel production.
These findings could herald a new age in sustainable energy solutions. Imagine a world where carbon neutrality is not a nebulous ideal but a tangible reality, driven by the effective conversion of waste into energy. Methanol synthesized from carbon dioxide could serve as a versatile energy source, powering vehicles, heating homes, and generating electricity. Furthermore, its high energy density stands out as a noteworthy characteristic, positioning it as a feasible alternative to traditional fossil fuels.
Insights with Implications Beyond Energy
Beyond its immediate applications, this groundbreaking research yields critical insights that could advance the chemical industry as a whole. By deciphering the unique molecular interactions that govern catalytic efficiency, scientists can create an avenue for innovations that transcend current limitations. Understanding molecular behavior at such a granular level will not only optimize existing catalytic processes but may also inspire entirely novel methodologies in material synthesis.
Baker optimistically states, “There is a treasure trove of knowledge awaiting us as we deepen our understanding of molecular chemistry.” With each new discovery, researchers inch closer to realizing environmentally conscious solutions that could pave the way for a globally sustainable future.
The implications of this research extend well beyond the mundane task of recycling waste molecules. Each new breakthrough represents a step towards changing which materials we deem waste, redefining our relationship with what we discard. In the realm of renewable energy, the far-reaching potentials of these findings offer encouragement that the pursuit of sustainability is not merely a lofty aspiration, but a vivid reality waiting on the horizon.

Leave a Reply