Recent advancements in the field of molecular and coordination chemistry have opened new avenues for redox chemistry, a process crucial in various scientific domains, including materials science and electrocatalysis. Led by Professor Ingo Krossing and his team at the University of Freiburg, this research pushes the limits of oxidation potential in positively charged ions. Traditionally, the oxidation potential for these ions reached only up to +0.65 to +1.0 volts versus the ferrocene standard (Fc+/0). The Freiburg team has remarkably elevated these potentials to +1.50 to +1.52 volts using novel methodologies. This article delves into the scientific techniques employed, the implications of these findings, and their significance in advancing chemical research.
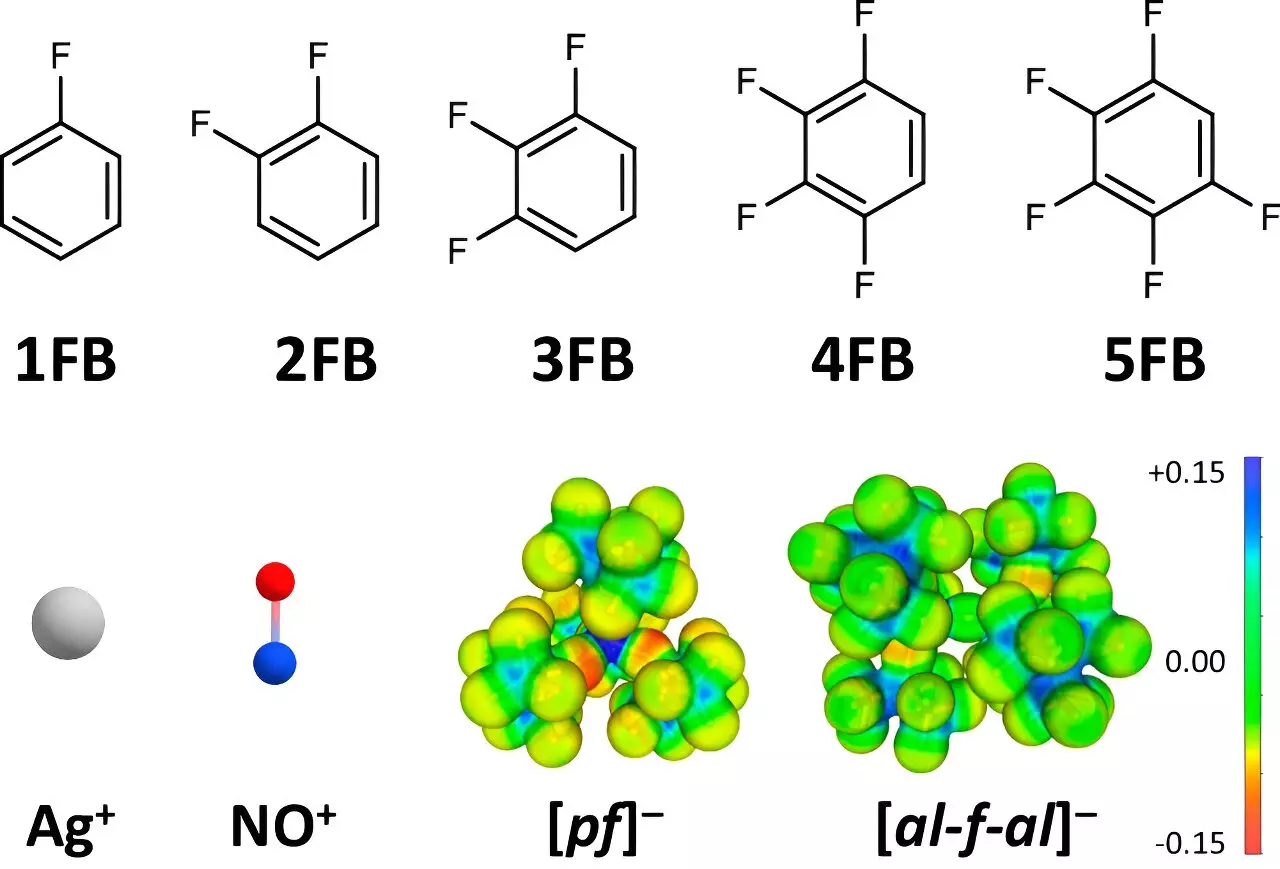
At the heart of this innovative research lies the strategic use of particular solvents and anions that are characterized by weak interactions with the ions being examined. While traditional solvents foster strong interactions that limit oxidation potential, Krossing’s team employed weakly coordinating anions (WCA) and selected fluorinated benzene derivatives as solvents. These carefully chosen components minimize the interaction between the solvent and the Ag+ and NO+ ions, effectively enhancing their oxidation capabilities.
Professor Krossing and his colleagues recognized that small, highly charged ions display strong interactions with their solvent environments, which in turn suppresses their oxidation potential. By utilizing specially modified fluorinated benzene derivatives, the team achieved a significant reduction of these interactions, thus enabling previously inconceivable oxidation reactions.
Understanding the physical properties of solvents is essential in determining their effectiveness in facilitating chemical reactions. Dr. Johannes Hunger from the Max Planck Institute for Polymer Research contributed significantly to this research by identifying critical dielectric constants of the chosen solvents. Results indicated that fluorinated aromatic compounds exhibited superior dielectric properties compared to common solvents like dichloromethane or acetone. This scientific insight into dielectric behavior underscores the pivotal role that solvent selection plays in enhancing the oxidation potential of positive ions.
The research team observed that as the level of fluorination increased, the interaction between the solvent and the cations decreased, thereby augmenting the oxidation potentials to unprecedented levels. Such findings contribute substantially to understanding how molecular design can impact reaction outcomes in inorganic chemistry.
The ramifications of this groundbreaking work extend beyond simple oxidation potentials. By demonstrating the feasibility of achieving higher oxidation capacities, the team forecasts vast potential applications in electrocatalysis and the development of redox shuttles or mediators. These findings open pathways to study previously hard-to-oxidize chemical compounds and may provide a framework for synthesizing new materials with enhanced properties.
The versatility of this research is also noteworthy. With a clear understanding of how solvent characteristics affect cation behavior, researchers can tailor experiments to explore complex molecules and reactions, driving innovation in fields such as renewable energy, battery technology, and even bioinorganic chemistry.
The work of Professor Krossing and his team was recently published in the esteemed journal Nature Communications, garnering attention for its implications in both theoretical and practical chemistry. As the scientific community begins to grasp the importance of these findings, there is an exciting potential for collaboration across disciplines to explore the untapped possibilities of enhanced oxidation potentials.
This research exemplifies how strategic molecular choices can significantly alter chemical behavior, reaffirming that the meticulous investigation of solvent interactions and ionic characteristics is a fruitful avenue in the realms of chemistry. With high oxidation potentials now within reach, a new era of chemical studies and applications appears on the horizon, paving the way for transformative advancements in scientific exploration.

Leave a Reply