Environmental challenges, particularly those posed by hydrocarbon pollutants, have long plagued industries and ecosystems alike. Traditional methods for degrading harmful hydrocarbons often fall short, either in efficiency or eco-friendliness. However, a remarkable breakthrough has emerged from the research team at UNIST, led by Professor Jaeheung Cho, who has harnessed the natural prowess of metalloenzymes to create a catalyst that promises a new era in pollution control. This innovative catalyst mimics the biological mechanisms found in nature, providing a more sustainable pathway to degrade harmful compounds in a less energy-intensive manner.
The Catalyst: Mechanism and Benefits
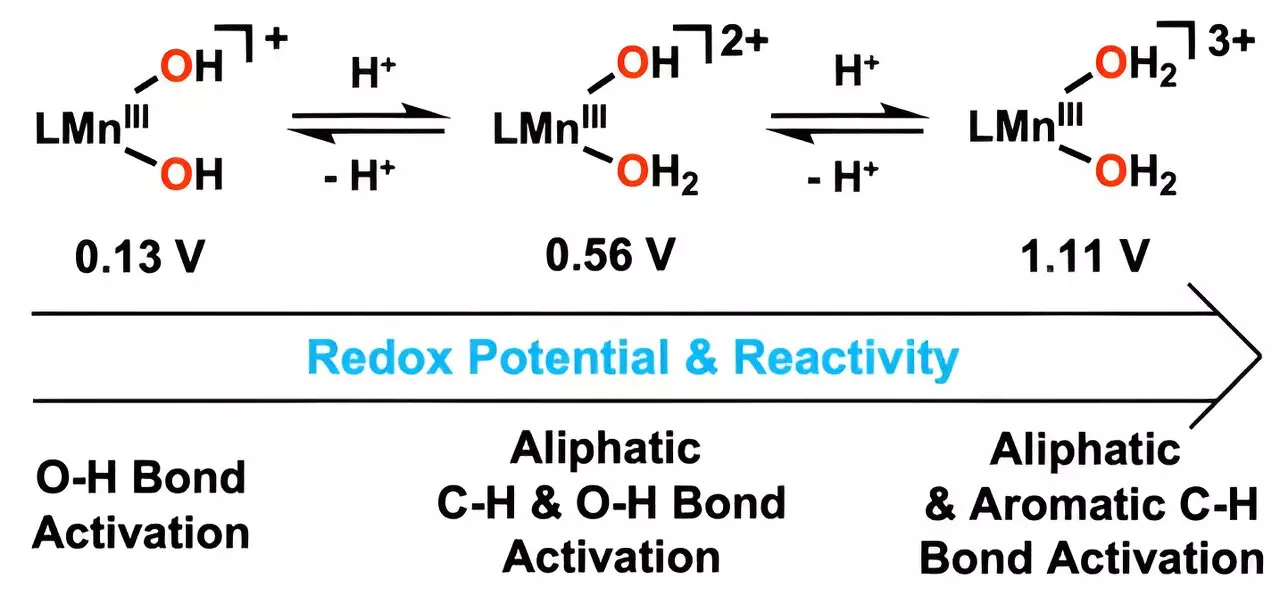
The newly developed catalyst operates through a sophisticated reaction mechanism, utilizing metal-bound water molecules to directly target carbon–hydrogen (C–H) bonds. Unlike previous techniques that require excessive heat or aggressive chemicals, this catalyst performs effectively at significantly lower temperatures. This shift could lead to a transformative reduction in energy consumption during pollution remediation processes, thereby not only addressing environmental issues but also easing economic burdens associated with high-temperature industrial operations.
The research team’s meticulous approach involved altering the hydroxyl ligand by integrating hydrogen ions, which boosted the catalyst’s function. By enhancing the reduction potential of manganese, they created a powerful catalyst that excels in activating oxygen–hydrogen bonds. This innovation is not just a scientific curiosity; it represents a practical tool that industries can adopt to tackle the pervasive problem of aromatic hydrocarbons, which are notoriously notorious for their toxicity and stability.
Groundbreaking Efficacy and Future Implications
Professor Cho’s team demonstrated the catalyst’s efficacy by successfully oxidizing anthracene, a particularly stubborn hydrocarbon, at low temperatures. The implications of this achievement are profound, opening new avenues for the breakdown of dangerous compounds once thought resilient to transformation. By focusing on aromatic hydrocarbons, the research aligns with urgent global goals for cleaner industrial practices and environmental sustainability.
Given that this is reportedly the first instance of a manganese(III) complex reacting efficiently with aromatic hydrocarbons at low temperatures, the potential applications of this catalyst extend well beyond laboratory confines. The breakthrough signifies a crucial step toward developing robust, industrially viable metal catalysts that could revolutionize pollution management across multiple sectors, including petroleum, manufacturing, and waste treatment.
Transforming Industrial Practices
In an age increasingly defined by the need for sustainable practices, the development of such innovative catalysts could redefine how industries approach hydrocarbon pollution. The implications extend to regulatory frameworks, consumer expectations, and corporate responsibilities, as companies could be motivated to invest in cleaner technologies that not only comply with environmental standards but also enhance their market competitiveness.
Ultimately, this revolutionary catalyst symbolizes a pivotal moment both in the field of chemistry and in broader environmental stewardship. It is a clarion call for researchers and industry leaders alike to leverage scientific ingenuity for the betterment of the planet. As the world grapples with the consequences of pollution, breakthroughs like this signal hope that we can still forge sustainable paths forward.

Leave a Reply