The phenomenon of self-organization has long fascinated scientists, acting as a fundamental characteristic that distinguishes living organisms from inanimate matter. The ability of biological systems to spontaneously form ordered structures is observed in a variety of contexts, from cell division to embryonic development. Recent research led by Professor Anđela Šarić at the Institute of Science and Technology Austria (ISTA) has shed light on an intriguing mechanism regarding bacterial cell division that encapsulates this paradigm. By investigating the behavior of the FtsZ protein—an essential component involved in bacterial cell division—Šarić and her team have discovered a previously unidentified process that illuminates the intricacies of life at a molecular level.
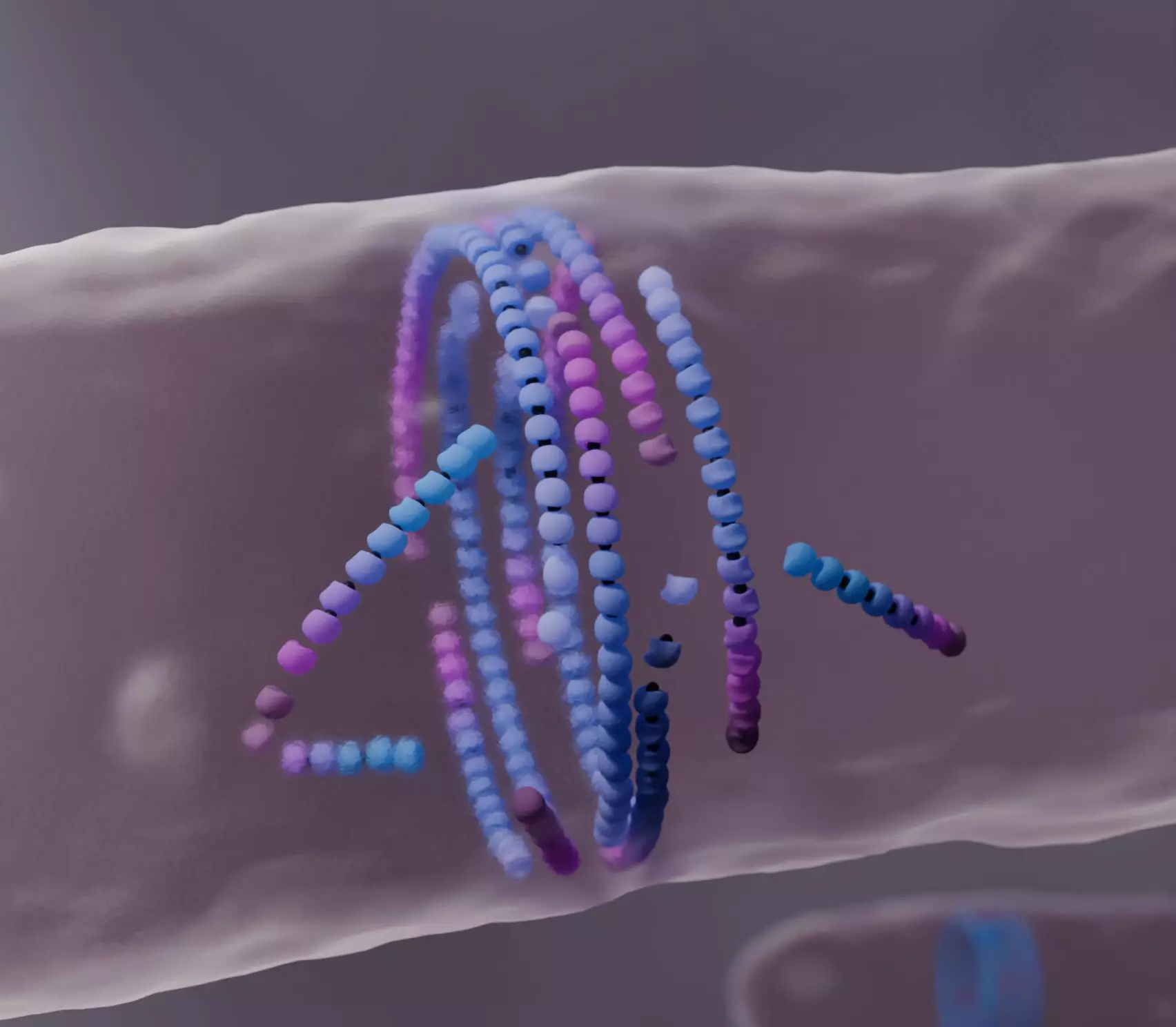
FtsZ is a protein that assembles into filaments within bacterial cells, forming a structure known as the division ring. This ring is crucial for the separation of daughter cells during mitosis. Traditionally, biological models have conceptualized FtsZ’s dynamics through the lens of ‘treadmilling,’ where subunits are continuously added to one end of a filament while being removed from the other. This process allows for a seemingly perpetual motion that is integral to cellular functionality. However, the research team sought to understand how these interactions contribute to the accurate alignment necessary for effective cell division, especially given the physical constraints present within the cellular environment.
The researchers approached the study of FtsZ with a fresh perspective by treating it as a type of ‘active matter’ that operates under different principles compared to passive systems. Their computational simulations revealed that misaligned FtsZ filaments “die” upon encountering obstacles, a phenomenon that might appear counterintuitive. Rather than pushing against misalignments, filaments dissolve and reassemble, effectively promoting alignment and forming a well-structured division ring. This unexpected behavior can be described by the phrase “dying to align,” as misalignment triggers a process that aids in achieving the desired structural configuration.
Collaborative Research: Bridging Theory and Experimentation
A key aspect of the Šarić group’s research is the successful collaboration with experimental teams, notably led by Seamus Holden from The University of Warwick and Martin Loose at ISTA. Their combined expertise allowed for a multifaceted approach that strengthened both the theoretical models and experimental validation. At a conference aptly named “Physics Meets Biology,” live imaging data presented by Holden illustrated the dynamic processes of FtsZ filament formation in real-time, mirroring the computational results from Šarić’s simulations. This cross-disciplinary engagement exemplifies how collaborative science can foster robust discoveries that bridge different fields of inquiry.
Applications Beyond Biology
While the immediate focus of this research lies in understanding bacterial cell division, the implications of these findings extend into the realm of synthetic materials. The mechanics of FtsZ filaments and their self-organizing capabilities present a potential framework for developing synthetic self-healing materials—substances capable of autonomously repairing themselves when damaged. This could revolutionize various industries, from construction to electronics, by integrating biological principles into material science.
Future Directions: A Quest for Living Matter
As the team continues to explore the fascinating world of active matter, they aspire to not only further elucidate the mechanics behind the formation of the bacterial division ring but also investigate its role in cell wall synthesis. The overarching goal of Šarić’s research is to innovate ways in which non-living materials can be engineered to mimic living processes, potentially leading to the creation of artificial cells or self-repairing technologies. By understanding the subtleties of molecular interactions, the researchers hope to unlock the secrets of life, shaping a new frontier in both biological and synthetic sciences.
The work spearheaded by Professor Anđela Šarić and her team at ISTA represents a significant advancement in our understanding of bacterial cell division and active matter dynamics. By demonstrating that misaligned FtsZ filaments aid in self-organization through a process of ‘dying to align,’ they redefine traditional notions of molecular behavior. Their research not only enhances our fundamental knowledge of biological systems but also spins a web of potential applications in synthetic material creation. As we continue to unravel the mysteries of life at its most basic structures, the convergence of biology, physics, and engineering paves the way for innovative solutions that could mimic life itself.

Leave a Reply